Products
Products
- OTC Products
Development and improvement of products that cater to the needs of customers
-
 In Japan, our main line of OTC products is external pain relieving drugs, including brands such as SALONPAS®,NOBINOBI® SALONSIP®, AIR® SALONPAS® and FEITAS®,as well as BUTENALOCK®, a remedy for athlete′s foot which contains components from prescription drugs approved for use in OTC products, and ALLEGRA®FX, a sinus medication for allergies, We also manufacture and sell a range of other products, such as quasi-drugs and cosmetics.
In Japan, our main line of OTC products is external pain relieving drugs, including brands such as SALONPAS®,NOBINOBI® SALONSIP®, AIR® SALONPAS® and FEITAS®,as well as BUTENALOCK®, a remedy for athlete′s foot which contains components from prescription drugs approved for use in OTC products, and ALLEGRA®FX, a sinus medication for allergies, We also manufacture and sell a range of other products, such as quasi-drugs and cosmetics.
In addition to developing and improving products that cater to the needs of customers, we take an active stance in marketing our products to increase our brand recognition through television and other media.
- Prescription Products
Earn the trust of customers by maintaining a high quality
-
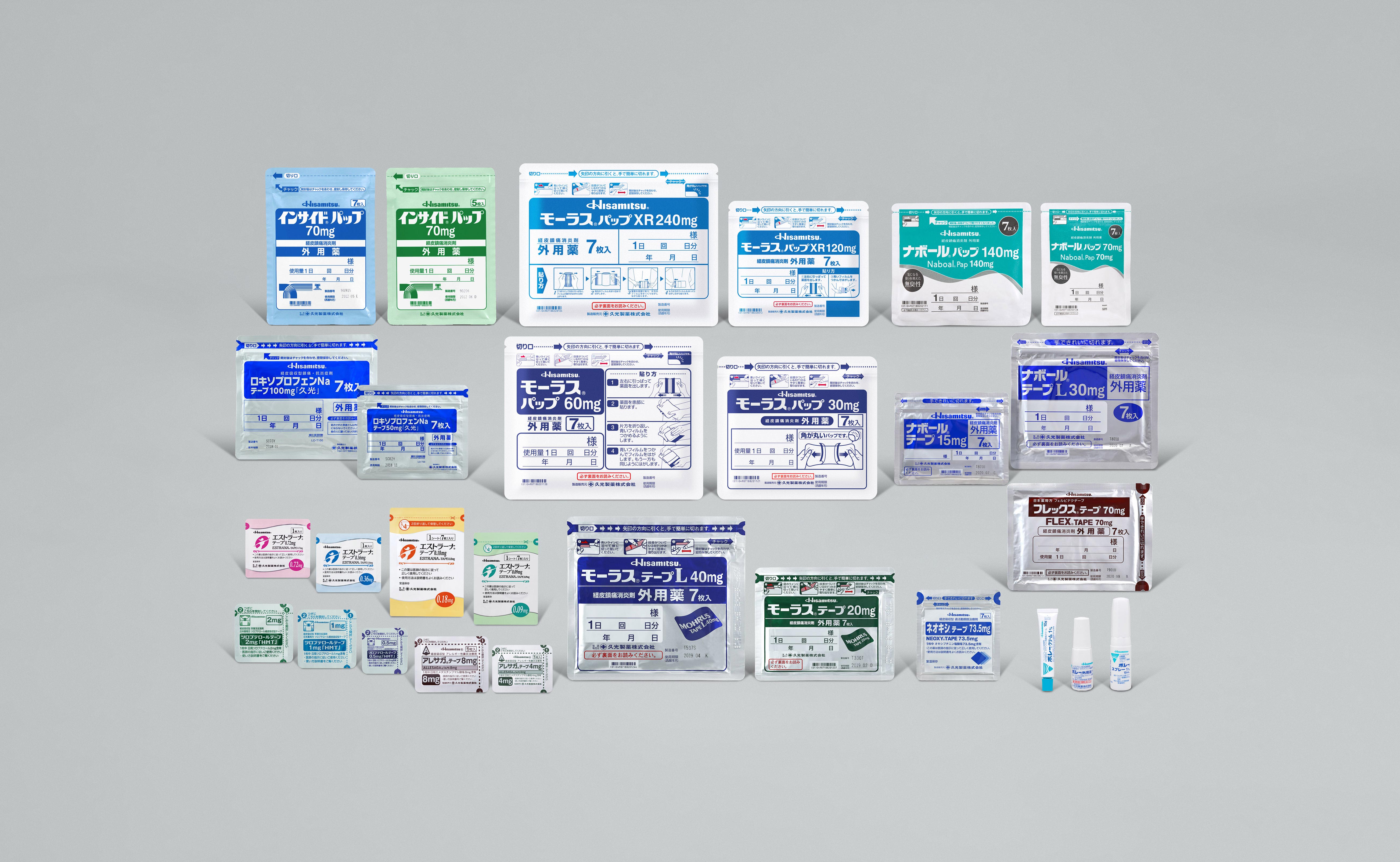 We manufacture and sell prescription products like MOHRUS® TAPE and MOHRUS® PAP XR, transdermal analgesic anti-inflammatory drugs. Other products we sell include female hormone patches, patches for asthma treatment and patches for treating overactive bladders.
We manufacture and sell prescription products like MOHRUS® TAPE and MOHRUS® PAP XR, transdermal analgesic anti-inflammatory drugs. Other products we sell include female hormone patches, patches for asthma treatment and patches for treating overactive bladders.
We also collect and supply information relating to the efficacy and safety of our products to medical facilities with the aim of providing new localized and systemic transdermal pharmaceutical products that meet the needs of patients.
- Export Products and Products manufactured outside of Japan
Delivering a better QOL to the world by providing high quality patches
-
 Our international operations, which began in 1937 with exports of the OTC product Salonpas®, have since expanded to cover more than 40 countries and regions, including Asia, the United States of America and the Euro area. On May 17, 2019, Salonpas® was recognized as the brand with the world’s No.1 market share in the category of OTC topical analgesic patches for the third consecutive year since 2016 by Euromonitor, a global market research firm. In the field of prescription drugs, we market a Ketoprofen containing patch similar to MOHRUS® TAPE in HongKong by the name of MOHRUS® PATCH. In Taiwan, we have acquired an approval of manufacturing and marketing permit for OABLOK® PATCH, a transdermal, overactive bladder treatment patch. Our international operations seek to improve our brand’s standing in terms of intellectual property, manufacturing technologies and quality control technologies while further enhancing our product plants and promote clinical trials.
Our international operations, which began in 1937 with exports of the OTC product Salonpas®, have since expanded to cover more than 40 countries and regions, including Asia, the United States of America and the Euro area. On May 17, 2019, Salonpas® was recognized as the brand with the world’s No.1 market share in the category of OTC topical analgesic patches for the third consecutive year since 2016 by Euromonitor, a global market research firm. In the field of prescription drugs, we market a Ketoprofen containing patch similar to MOHRUS® TAPE in HongKong by the name of MOHRUS® PATCH. In Taiwan, we have acquired an approval of manufacturing and marketing permit for OABLOK® PATCH, a transdermal, overactive bladder treatment patch. Our international operations seek to improve our brand’s standing in terms of intellectual property, manufacturing technologies and quality control technologies while further enhancing our product plants and promote clinical trials.

