Sustainability
Society
Bonds with Customers
Provide Value for Society, Using High Quality Pharmaceutical Products and Services Backed by Unique Technology
Since the development of SalonpasⓇ, the Hisamitsu Pharmaceutical Group has continued to develop and improve our products by promptly responding to customer needs. At the same time, Hisamitsu has refined the TDDS technology (including the Transdermal Patch Technology), to create a variety of products such as Rx drugs, OTC drugs, and skincare products. Going forward, we will not only spread the “Patch Treatment Culture” of our transdermal patches but will also promote the “TE-A-TE” culture worldwide and conduct research and development on a global platform to deliver products that meet the needs of our customers in a timely manner. Constantly thinking about “Delivering a Better QOL to the World” with products that embody the spirit of “TE-A-TE,” we shall provide more effective and safer products and services to the world.
Value to Be Presented to Society
Since our establishment, we have been engaged in product development to satisfy customers’ needs in accordance with the basic policy of “The Customer-first Principle,” with our main focus on transdermal patches through which pharmaceuticals can be administered on a stable basis and which can be patched with comfort. In addition, we promptly reflect customer needs into our products and quickly respond to changes in the social environment, such as the aging population. To do so, we conduct product sampling to let customers and medical workers directly know about the excellence of our products, and also ensure cooperation between the actors in our value chain. While continuing to create value that will contribute to improving the environment and society, we will achieve global application of our technology developed in Japan so that we can present the world with new value that will contribute to citizens’ healthy and rich lives.
R&D
Based on the fundamental technology platform for TDDS that we have developed over many years, the R&D Division is working to develop TDDS formulations of drugs that are difficult to absorb through the skin. We are engaged in the research, development, and improvement of drugs that are clearly differentiated from existing and competing products by maximizing the benefits of TDDS formulations, such as stable efficacy and reduced adverse drug reactions resulting from sustained drug concentrations in the blood.
Additionally, in order to launch new pharmaceutical products in the global market as quickly as possible, we are striving to consolidate our R&D efforts made at our domestic R&D sites and our overseas subsidiary (Noven Pharmaceuticals, Inc.) and shorten the development pipeline period.
Strengthening Our R&D Organization
The R&D Division’s primary mission is to launch new products and enhance the value of existing products (through additional indications, product improvements, etc.) To achieve the Seventh Medium-term Management Policy and ensure the sustainable growth of the Hisamitsu Pharmaceutical Group, we have restructured our existing R&D organization into a Group R&D organization centered around the SAGA Global Research Center. We also work closely with the pharmaceutical affairs department to ensure the smooth development of Rx drugs overseas.
In addition to reorganizing the research department into five units, we have made progress in establishing a comprehensive R&D infrastructure, including actively promoting open innovation at the SAGA Global Research Center, collecting the latest information at Shonan Health Innovation Park, and collaborating with Noven Pharmaceuticals in responding to FDA (U.S. Food and Drug Administration) inquiries.


| Status Quo in FY2024 | Challenges to Be Addressed | Major Stakeholders | Actions in FY2025 |
|---|---|---|---|
| Progress in overseas expansion |
Taking measures to comply with relevant systems in overseas countries where we operate |
Relevant administrative authorities, healthcare professionals |
Building relationships with related organizations Collaborating with related departments within the Company |
| Strengthening cooperation among group personnel | Employees | Enhancing communication among group researchers | |
| Enhancement of pipelines |
Advancing open innovation | Partners, employees | Revitalizing the SAGA Global Research Center Searching for and strengthening collaboration with research institutions and ventures |
Development of a New R&D Organization
The main mission of R&D Division is to bring new products into the market and to develop existing products (additional indication, product improvement). In order to achieve the Seventh Medium-term Management Policy and solidify the future potential of Hisamitsu Group, we are restructuring the former R&D organization into a Group R&D organization centered on SAGA Global Research Center.
For the R&D organization, we reorganized the former research departments into five units. With this reorganization, we will promote R&D more efficiently, ranging from basic research to development and tests, including support for various modalities.
For global expansion of the Group, we will seek to promote flexible development in line with the trend and legal restrictions of each country or market, mainly led by the SAGA Global Research Center.
In addition, we have also established a structure for managing intellectual properties strategically by proceeding with collaboration with parties concerned at Shonan Health Innovation Park and promotion of open innovation.
5 units for Enhancing R&D

Drug Development
The Hisamitsu Pharmaceutical Group is committed to the research and development of TDDS formulations to fulfill the fundamental desire for “health, safety, and comfort for people around the world.” Topical formulations, such as patches, gels, and sprays, not only maintain stable drug blood concentrations over extended periods, but also have the advantage of being suitable for administration to patients with swallowing difficulties, such as children and the elderly, and allowing easy discontinuation if side effects occur. This allows us to meet a wide range of medical needs, regardless of clinical specialty. In our research and development, we strive to develop highly useful TDDS formulations through exhaustive searches for candidate drugs and active promotion of alliances. Furthermore, we actively use new fundamental technologies such as microneedles to develop TDDS for a wider range of drugs, including macromolecular drugs and vaccines, in order to develop drugs that meet the needs of the medical field.
“Promoting ‘TE-A-TE’ Culture Worldwide” with TDDS Technology
Microneedles are attracting attention as a new technology in TDDS Development Improvement (transdermal drug delivery system). They are a groundbreaking drug delivery system with an immediate effect and reduced invasiveness at the time of administration, something that has been difficult to achieve with conventional dosage forms. In addition to using the properties of microneedles to expand into new therapeutic areas, we are also working to expand into the cosmetics field by applying a pleasant skin stimulation sensation. We are currently preparing to commercialize new products, with a view to partnering with other companies. We are also continuing to offer traditional patches to address a variety of illnesses. Specifically, we aim to offer a new lineup following products such as APOHIDEⓇ Lotion, a treatment drug for primary palmar hyperhidrosis; ZICTHORUⓇ Tapes, a transdermal analgesic for persistent pain; and HARUROPIⓇ Tapes, a transdermal dopaminergic Parkinson’s disease treatment. At the same time, we value the spirit of “TE-A-TE” and are exploring the development of a variety of products and services that contribute to improving people’s quality of life (QOL). As part of this, we have begun venturing into new areas, such as health foods and therapeutic apps. In the development of Rx drugs in the U.S., to accelerate the overseas expansion of ZICTHORUⓇ Tapes, we are advancing the clinical development of HP-3150 (generic name: diclofenac sodium), an analgesic to meet the indications of chronic lower back pain, replacing HP-5000 (generic name: diclofenac sodium), a transdermal analgesic and anti-inflammatory drug for the indications of knee osteoarthritis. In addition, sales of XELSTRYMⓇ (development code: d-ATS, generic name: d-amphetamine), a transdermal treatment drug for attention-deficit hyperactivity disorder, have steadily increased since its launch in the U.S. in 2023, achieving a significant increase in FY2025 compared to the previous fiscal year.
R&D Pipeline
Click here for information on our R&D pipeline.
Expansion of Target Diseases for Hisamitsu TDDS
Microneedle Technology
HalDiscⓇ Technology
(Pinhold-shaped microneedle technology)
Realization of macromolecular drugs that are treated as injectable drugs such as vaccines, offering easy self-medication by anyone
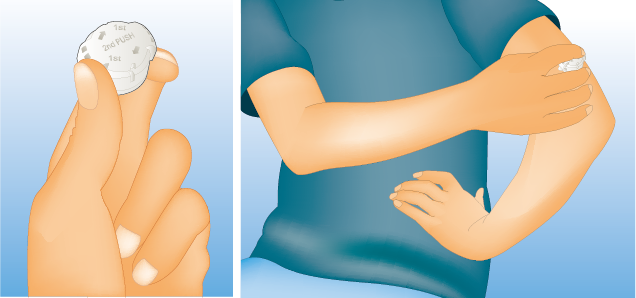
SheetifyⓇ Technology
(Sheet-type microneedle technology)
Hisamitsu's proprietary sheet-type device for the realization of high-dose transdermal administration
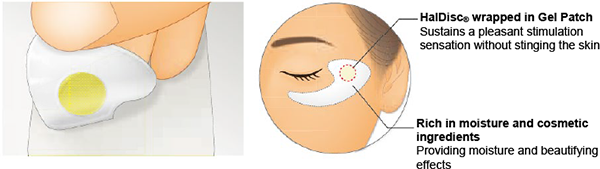
HalDisc beauteⓇ
(Cosmetic Microneedle Technology)
Hybrid technology for cosmetic use that combines transdermal absorption technology and microspike substrates to create a pleasant stimulation sensation
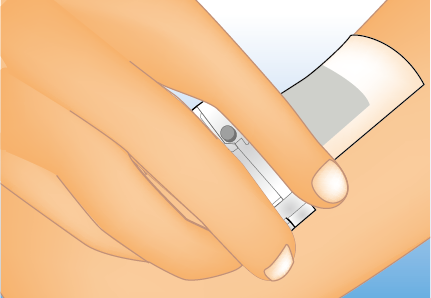
HP-6050 (microneedle formulation)
Using the benefits of HalDiscⓇ Technology, which allows for safer and easier transdermal drug administration compared to conventional injections, this drug is currently being developed as a treatment for sedation procedures for patients with delirium, psychomotor excitement, and irritability. It is expected to contribute to reducing the burden on both healthcare professionals and patients by reducing the need for restraints to ensure safety during procedures. If approved, this drug will be the world’s first microneedle formulation as an Rx drug.

Drug Improvement
When improving our drugs, we focus primarily on customer feedback (needs), the latest manufacturing technologies, and the results of research and development. Aiming to improve our customers’ QOL, we are continually working to select the size and shape of formulations and reduce the number of times they are used.Until now, we have focused on the perspectives of the manufacturer and the user. However, as the Hisamitsu Pharmaceutical Group works to promote ESG and achieve the SDGs, we believe it is important to actively incorporate new perspectives. Specifically, we will actively work to improve our products to make them ecofriendly, with the aim of reducing CO₂ emissions and the impact on resource recycling. As one measure to reduce environmental impact, we have established a subcommittee within the Sustainability Promotion Committee to focus on reducing the amount of plastic used in our products, with the R&D and production departments playing a central role in promoting this initiative. Furthermore, from the perspective of open innovation, we promote collaboration with suppliers and use a wide range of technical information.
Consideration for the Packaging of Rx Drugs
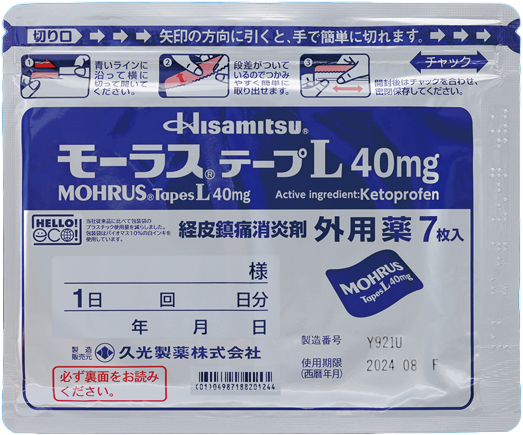
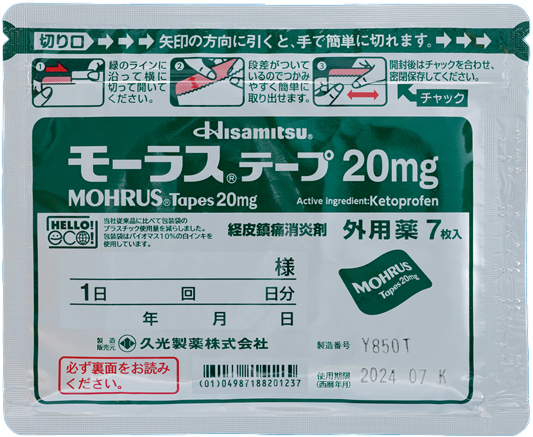
We recognize that environmental consideration is an important management issue for drugs in general, especially for Rx drugs. Hisamitsu Pharmaceutical is continuously developing and improving drug packaging with environmental impact in mind.As one of our achievements, in 2023, the packing pouch for MOHRUS® Tapes 20 mg and MOHRUS® Tapes L 40 mg won the Optimum Packaging Award of Japan Packaging Contest hosted by the Japan Packaging Institute and the Packaging Technology Award of the Kinoshita Award. This was in recognition of our achievement in reducing environmental impact while maintaining the same quality as conventional products.Going forward, we will continue to contribute to the realization of a sustainable society by striving to balance stable drug supply with environmental protection through the following initiatives.
- ●Promoting the use of recycled PET
- ●Reducing resource consumption by thinning packing pouchesthrough changes in material composition
- ●Reducing energy consumption in production processes
- ●Reducing plastic usage and waste
Kinoshita Award:This is an award program established to commemorate the achievements that the late Mr. Matasaburo Kinoshita, the second chairman of the Japan Packaging Institute, who has been accomplished in the packaging industry for many years.
Japan Packaging Contest:This competition selects the best products (Good Packaging Award) of the year based on all aspects including materials, specification, technology, appropriate packaging, environmental friendliness, design, transport packaging, logistics, sales promotion, and ideas.


Ethical Drug Development
Ethical and Scientific Clinical Studies
The Hisamitsu Pharmaceutical Group places the highest priorityon ensuring the human rights and safety of patients participating inclinical trials. To that end, we prepare clinical trial protocols in strictcompliance with the Act on Pharmaceuticals and Medical Devicesand Good Clinical Practice (GCP), and always obtain informedconsent from patients.We have also established the Internal Institutional Review Board,which includes outside medical experts, to rigorously assess theethical and scientific validity of clinical trials. In overseas clinical trials,we comply with ICH-GCP (guidelines of the International Council forHarmonisation of Technical Requirements for Pharmaceuticals forHuman Use),* and conduct them in accordance with the regulatoryrequirements and guidelines of each country and region.We recognize that ethical societal demands in drug developmentwill continue to grow in the future, and we believe it is important toconstantly keep global trends and public awareness in mind andrespond appropriately.
* ICH-GCP : International GCP Guidelines on the practice of clinical trials and clinical studies agreed at the Japan-U.S.-EU ICH (International Council for Harmonisation of Technical Requirements for Pharmaceuticals for Human Use)
Ethical Accommodation for Animal Welfare
Hisamitsu Pharmaceutical has formulated the Animal Testing Guiding Principle in line with the Act on Welfare and Management of Animals to ensure that respect for animal welfare is completely shared and that animal testing is performed properly with accommodation for animal welfare. For animal testing to inspect drug efficacy and safety, we have a system requiring all such testing to be reviewed by the Animal Testing Committee, and research is conducted under the 3R Principles: Replacement, Reduction, and Refinement. In addition to regular self-inspections, our animal testing operations underwent third-party review, and in July 2025, the SAGA Global Research Center gained accreditation as an animal testing facility.
Procurement
The procurement department not only ensures the quality andstable procurement of raw materials, reduces costs, and adheresto delivery deadlines, but also strives to improve our supply chainand collect and share information from suppliers internally. AtHisamitsu Pharmaceutical, the procurement department and relateddepartments work together from the product planning and researchstages to discuss raw materials, thereby building a stable supplysystem and enabling product development that quickly meetscustomer needs. We are advancing initiatives in accordance with the SustainabilityProcurement Basic Policy and Sustainability Procurement Standardsfor Suppliers, which we established as our Sustainability ProcurementGuidelines in 2021. Furthermore, in 2023, we published theDeclaration of Partnership Building. We aim to build new partnershipsby promoting collaboration, coexistence, and mutual prosperity withsupply chain partners and businesses seeking to create value. We notonly comply with the laws and regulations of each country and region,but also address human rights risks and halal issues in the supplychain, and select appropriate raw materials allowing people in manycountries can use our products with peace of mind.
Declaration of Partnership Building

This time, we announced the “Declaration of Partnership Building” on July 26th, 2023, in agreement with the aims of the “Council for the Building Partnerships to Lead the Future”, which is promoted by the Cabinet Office and The Small and Medium Enterprise Agency. We aim to build new partnerships by promoting cooperation and mutual prosperity with our suppliers in the supply chain and with businesses that create value.
Production
With our two GMP-compliant factories, Tosu Factory (SagaPrefecture) and Utsunomiya Factory (Tochigi Prefecture), positionedas the base of domestic production, we have built a stable productsupply system that also includes our overseas factories, workingto improve productivity every day. Within the Production Division,we have created a cross-sectional team to continuously carryout KAIZEN activities aimed at improving quality and productivity.Furthermore, to promote the global horizontal expansion of KAIZENactivities, we hold the Hisamitsu KAIZEN Awards, with the aim ofraising awareness of KAIZEN activities among employees, includingthose at overseas subsidiary factories. We hope that these activitieswill inspire factories in each country and create technologicalcompetition, further developing the Hisamitsu PharmaceuticalGroup.
*GMP:Good Manufacturing Practice
Logistics
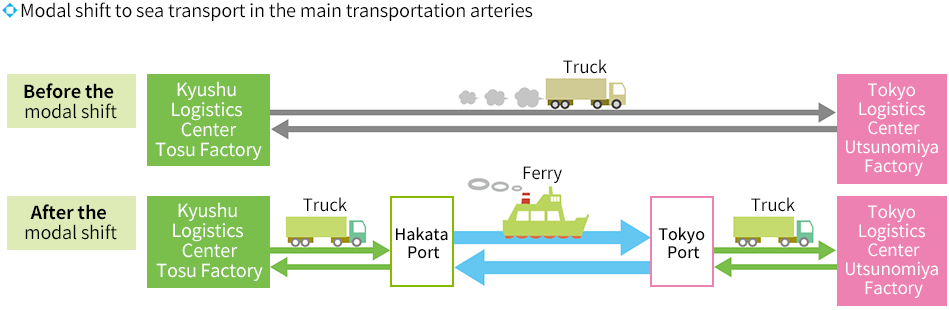
In addition to our domestic logistics centers in eastern Japan(Kuki City, Saitama Prefecture) and western Japan (Tosu City,Saga Prefecture), we established the Kansai Logistics Center(Kumiyama Town, Kuze County, Kyoto Prefecture) in April 2024,creating a logistics system that can ensure a more stable supply ofpharmaceuticals. Each center has supervising pharmacists assigned to ensurepharmaceutical quality control, and information on shipping volumeis shared with factories. This allows us to properly store and managea wide variety of pharmaceutical products and ensure a stable supplyto our customers. We also actively work to improve transportationefficiency and reduce our environmental impact. In addition to ourcurrent truck transport, we are implementing a modal shift to railand sea transport, which have a lower environmental impact. Wewill strive to further improve transportation efficiency by expandingmodal shifts and improving loading efficiency on a continual basis.
Modal Shift to Rolling Stock and Sea Transportation as the Main Transportation Channel
Sales/Marketing/Service
At sales/marketing/service, we provide and collect information on the effectiveness and safety of our products to enable medical workers and patients to use them with peace of mind and also to ensure appropriate use of the products. Additionally, requests from healthcare professionals collected by MRs are shared with related departments to further improve products, and the OTC business also promotes “Jissen” in which samples are distributed to directly communicate the superiority of products to consumers.
We help not only patients in the medical field, but also other people around the world maintain and improve their good health.
Japan - Rx business
Continued Provision of Information related to Primary Palmar Hyperhidrosis
While primary palmar hyperhidrosis was often dismissed as anuntreatable condition, awareness that it is a treatable disease isnow steadily spreading not only among patients themselves butalso among healthcare professionals and those around them. Wefeel that this has made a significant contribution to improving thequality of life (QOL) of many patients.However, awareness of the disease in society as a whole isstill insufficient. It is necessary for more patients and healthcareprofessionals to deeply understand that this disease is treatable andthat such treatment contributes greatly to improving patients’ QOL.For this reason, Hisamitsu Pharmaceutical continues to provideinformation from various angles. We aim to raise awareness not onlyamong patients but also among society as a whole through diseaseawareness activities using TV commercials, web commercials, andonline public lectures.We also strive to provide high-quality information to healthcareprofessionals by establishing an educational system for medicalrepresentatives (MRs) so that they can provide more appropriateinformation. Furthermore, we work to deepen awareness andunderstanding of the disease by holding study sessions in whichhealthcare professionals can experience the symptoms of primarypalmar hyperhidrosis.

Initiative to support continued treatment for patients suffering from sweaty hands



Japan - OTC Business
Promotion of Ethical Measures such as Zero Returns
Ethical consumption, which emphasizes consideration of ethical aspects such as environmental issues and human rights, has gained momentum in recent years, and its importance and ripple effects are spreading through media and social media. Amid this social trend, Hisamitsu Pharmaceutical has proactively addressed the issue of product returns, which has long been a major issue in the OTC business.Returns in the pharmaceutical industry are a complex issue stemming from business practices not only at pharmaceutical companies but also across the entire market, including retailers and wholesalers. However, in FY2024, we achieved the milestone of zero returns by promoting collaboration across the entire value chain, building on our strong partnerships with wholesalers and retail distributors. This demonstrates our contribution to a sustainable society by eliminating resource waste and improving efficiency throughout the supply chain. Going forward, we will further develop our existing “HELLO! eco!” mark initiatives and further strengthen our ethical business activities. We will also proactively disclose information about these achievements and deepen our communication with society.




The Importance of Human Resource Development in Light of Future Developments
To quickly grasp market changes and implement appropriatecountermeasures from a global perspective, it is essential to developthe necessary human resources to support this effort. HisamitsuPharmaceutical believes that it is important to foster a balance ofdigital talent who are responsible for disseminating information andusing big data, specialists who build strategies based on logisticsand information analysis, and generalists who function as a team. In human resource development, it is essential to implement acomprehensive training program that not only enables employeesto acquire various skills, but also fosters greater awareness ofembodying the Corporate Philosophy of “Delivering a Better QOL tothe World.” It is also important to increase employees’ experiencethrough appropriate job rotation.At the same time, we must address regional differences, such asurban concentration, and improve our work environment, focusingon work-life balance and diversity. Through these measures, wewill promote human resource development that balances individualgrowth with the growth of the organization as a whole.




Overseas - Rx and OTC Business
Building Global Partnerships
As we expand our business overseas, we recognize that buildingpartnerships in each country is even more important than in Japan.To date, we have invited distributors from around the world toJapan to deepen their understanding of the “TE-A-TE” culture,which Hisamitsu Pharmaceutical has cultivated over the years. Wehave also built strong relationships of trust through tours of ourfactories, which ensure the quality of our products, and throughmutual exchanges. In addition to these efforts, starting in 2023, wehave held an annual event for distributors from around the world toshare and gain understanding of the current status and directionof our OTC and RX drugs. This has also enabled lively and fruitfulexchanges of views about future business development in eachcountry, helping to promote our global business expansion.
We will continue to strengthen these global partnerships.



Strengthening Our Global Network
For Hisamitsu Pharmaceutical to make further strides overseas, itis essential to establish a stable supply system and a solid salessystem.In North America, where steady progress is expected, furtherstrengthening the sales system at Hisamitsu America and Novenand enhancing cooperation on the supply side will be key.In addition, we will strengthen the supply capacity of our threeoverseas subsidiaries with production sites: Hisamitsu Vietnam,Hisamitsu Indonesia, and Hisamitsu Brazil. At the same time, we willenhance our expansion into Asia and the Global South by improvingour sales infrastructure in existing countries and newly developedcountries in the Asia-EMEA region.*We will continue to strengthen our existing sites and build aglobal network of supply and sales systems with clear positioningaccording to the characteristics of each area, thereby contributingto improving the QOL of people around the world.*Three regions: Europe, the Middle East, and Africa

(New York, U.S.)

(Ho Chi Minh City, Vietnam)

Halal* Certification
Hisamitsu Pharmaceutical operates businesses in Indonesia, Malaysia, and other countries and regions where many Muslims live.In our subsidiary Hisamitsu Indonesia, in order to ensure that the haral guidelines in Indonesia are followed, and Muslims can use our products with peace of mind, we have established a Halal team within the Company and are promoting the creation of products that consider religion and the lifestyle of region.*Halal: What is legal under Islamic law

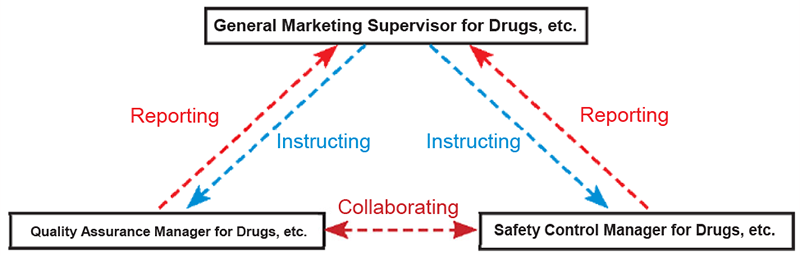
Quality Assurance
Quality is assured through GMP-compliant and scientifically established methods of production to ensure the reliability and safety of drugs.
Our quality assurance operations range extensively from “preparing process charts related to production and quality,” to “inspecting, testing and evaluating products,” “analyzing defect trends,” “auditing and instructing business partners,” and “correspondence with customers after shipment.” Furthermore, we carry out a wide range of internal and external quality assurance, including the creation of a smooth transition from the formulation development stage to the production stage to ensure quality assurance structure and management and auditing of the production system of contractors that manufacture OEM (*) products.
We have established an internal compliance promotion organization to monitor compliance with strict laws and ordinances and voluntary regulations. Additionally, we are constantly striving to improve quality and safety of our drugs under the strict quality control system, including inspections of our quality system by public agencies.
*OEM: Original Equipment Manufacturer
Commitment to Product Reliability Assurance
Since pharmaceuticals are life-related products, strict management standards are established to ensure their reliability, including GLP*1 and GCP*2 at the research and development stage, GMP*3 at the manufacturing stage, and GPSP*4 and GVP*5 at the post-marketing stage. Our Quality Assurance & Pharmacovigilance Division conducts rigorous audits focusing on control standards at each stage of product development, ultimately ensuring product reliability through a system of reliability assurance covering the entire product lifecycle. Furthermore, in line with the globalization of the Company, we are working to strengthen our auditing system by expanding the scope of risk assessment as well as complying with the latest regulations and guidelines in each country.
*1 GLP:Good Laboratory Practice Standards for practice of nonclinical studies for the safety of drugs.
*2 GCP:Good Clinical Practice Standards for practice of clinical studies of drugs.
*3 GMP:Good Manufacturing Practice Standards for production management and quality control of drugs and quasi-drugs.
*4 GPSP:Good Post-marketing Study Practice Standards for post-marketing research and practice of study on drugs.
*5 GVP:Good Vigilance Practice Standards for post-marketing safety management of drugs, quasi-drugs, cosmetics, medical devices and regenerative medicine.
Supplier Audits
In FY2024, we conducted written, online, and on-site audits of more than 50 raw material suppliers and product manufacturing contractors in Japan and abroad to confirm that manufacturing and quality control of raw materials and contracted products were being performed properly. The audit items are set mainly to cover important matters related to the operations of drug substance, raw material, and contracted product manufacturing plants; testing facilities; and other facilities based on GMP and other standards. Additionally, in the event of quality-related changes or complaints, we conduct a special audit to ensure product quality and stable supply.
Inquiries from Customers
At Hisamitsu Pharmaceutical, we have the Customer Center as a contact point for customers to make inquiries about our products. We respond to not only inquiries through our website, but also inquiries from individual customers by phone and letter.
While striving to make attentive, prompt, and appropriate responses to such inquiries, we share feedback and indications from customers internally among related departments in order to help improve the relevant products or develop new products.
